Multi-Therapeutic Network
of Clinical Research Sites
DM Clinical is a network of clinical trial investigator sites. For 18 years we have been connecting patients with our trusted physicians to conduct cutting-edge studies focused on quality and compassion.
Accelerate your trial journey
from site selection to close-out
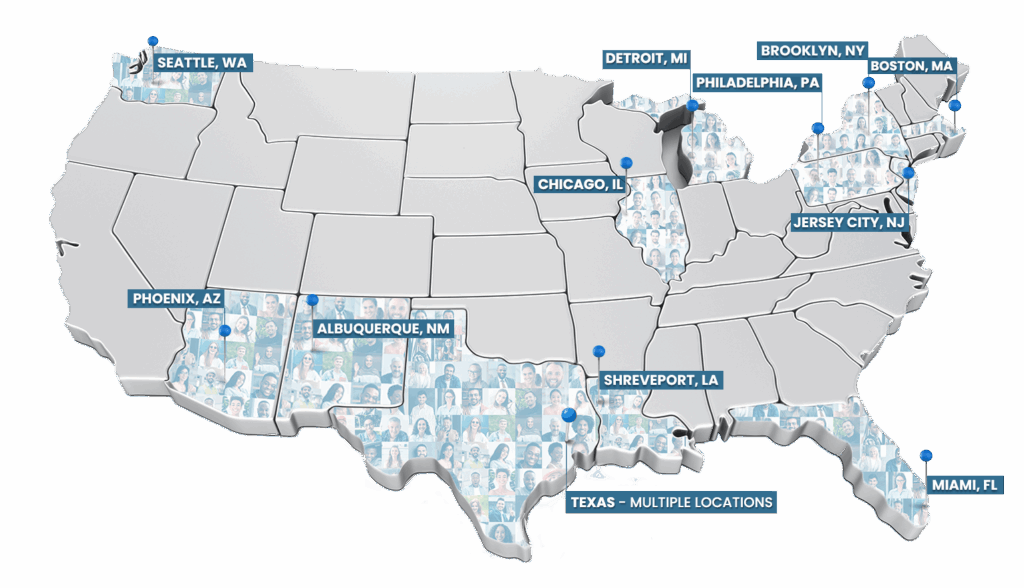
Research sites across the U.S.
Subjects Enrolled
Patient Database

MULTI-SPECIALITIES
Therapeutic Expertise

Infectious Diseases
HIV, Flu, Chickenpox, MMR, HPV, HiB, Hepatitis B, Pneumonia, C. Difficile, RSV, Pertussis, DTaP/TDaP, Meningitis

Infectious Diseases
HIV, Flu, Chickenpox, MMR, HPV, HiB, Hepatitis B, Pneumonia, C. Difficile, RSV, Pertussis, DTaP/TDaP, Meningitis

Gastroenterology
Crohn’s Disease, Ulcerative Colitis, Colonoscopy Studies

Pediatrics
Vaccines, Migraine, Asthma, Influenza, RSV
Pulmonology
Asthma, COPD, Chronic Respiratory, Lung

Psychiatry
psychiatry Psychiatry Schizophrenia, OCD,
Why participate in a clinical trial?
This is where medicine begins.
All because of people like you.
By joining a clinical trial, you can help scientists research potentially life-changing medications, vaccines, medical devices, and more. All clinical trials conducted at DM Clinical are overseen by the U.S. Food and Drug Administration (FDA) and led by licensed healthcare professionals.
Frequently Asked Questions
Learn More About Clinical Research.

Almost anyone can participate in a clinical trial. You can participate in clinical trials to advance medical research—regardless of your age, health, ethnicity or background. There is no insurance required, nor any proof of legal status or citizenship.
Yes. Every study is a little bit different.
Most trials offer compensation for your time and effort, which could be anywhere from a few hundred to a thousand dollars or more.
The compensation you receive is approved by an independent ethics board, and is relative to what is required of you in that trial—including the length and number of study visits. The compensation is detailed in the informed consent document you will receive at your first study visit.
When you speak with our Patient Enrollment Specialists, please feel free to discuss the compensation with them. We appreciate the sacrifice of your time and effort entailed in study participation, and want to be upfront with you about compensation.
It varies by study. Studies typically require you to come to the clinic a few times, complete e-diaries, and/or speak with clinicians on the phone. The majority of our trials are conducted in outpatient clinics—that means that no overnight stay is needed for most of the studies. Most visits are similar to what you would experience at your regular care visits to your doctor, such as a physical exam, vitals, medical history, blood/urine sample.
Participant Feedback on Trials
Reveal the information of commenters with text of their feedback. Use customer testimonials to convince the audience to perform the action.




